The Beach Boys’ iconic hit single “Good Vibrations” takes on a whole new layer of meaning thanks to a recent discovery by Rice University scientists and collaborators, who have uncovered a way to destroy cancer cells by using the ability of some molecules to vibrate strongly when stimulated by light.

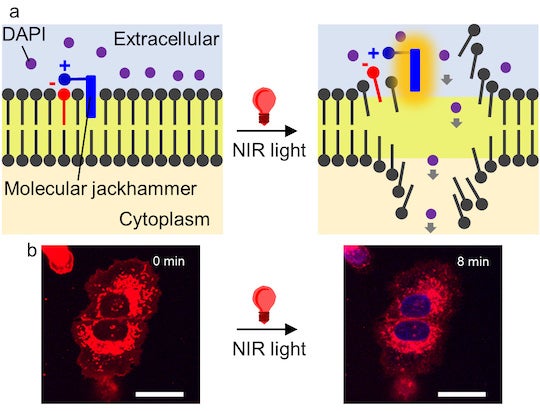
The researchers found that the atoms of a small dye molecule used for medical imaging can vibrate in unison ⎯ forming what is known as a plasmon ⎯ when stimulated by near-infrared light, causing the cell membrane of cancerous cells to rupture. According to the study published in Nature Chemistry, the method had a 99 percent efficiency against lab cultures of human melanoma cells, and half of the mice with melanoma tumors became cancer-free after treatment.
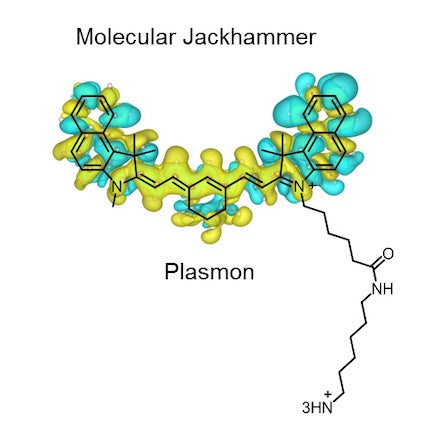
“It is a whole new generation of molecular machines that we call molecular jackhammers,” said Rice chemist James Tour, whose lab has previously used nanoscale compounds endowed with a light-activated paddlelike chain of atoms that spins continually in the same direction to drill through the outer membrane of infectious bacteria, cancer cells and treatment-resistant fungi.
Unlike the nanoscale drills based on Nobel laureate Bernard Feringa’s molecular motors, molecular jackhammers employ an entirely different ⎯ and unprecedented ⎯ mechanism of action.
“They are more than one million times faster in their mechanical motion than the former Feringa-type motors, and they can be activated with near-infrared light rather than visible light,” Tour said.
Near-infrared light can penetrate far deeper into the body than visible light, accessing organs or bones without damaging tissue.
“Near-infrared light can go as deep as 10 centimeters (~ 4 inches) into the human body as opposed to only half a centimeter (~ 0.2 inches), the depth of penetration for visible light, which we used to activate the nanodrills,” said Tour, Rice’s T. T. and W. F. Chao Professor of Chemistry and a professor of materials science and nanoengineering. “It is a huge advance.”
The jackhammers are aminocyanine molecules, a class of fluorescent synthetic dyes used for medical imaging.
“These molecules are simple dyes that people have been using for a long time,” said Ciceron Ayala-Orozco, a Rice research scientist who is a lead author on the study. “They’re biocompatible, stable in water and very good at attaching themselves to the fatty outer lining of cells. But even though they were being used for imaging, people did not know how to activate these as plasmons.”
Ayala-Orozco initially studied plasmons as a doctoral student in the research group led by Rice’s Naomi Halas.
“Due to their structure and chemical properties, the nuclei of these molecules can oscillate in sync when exposed to the right stimulus,” Ayala-Orozco said. “I saw a need to use the properties of plasmons as a form of treatment and was interested in Dr. Tour’s mechanical approach to dealing with cancer cells. I basically connected the dots.
“The molecular plasmons we identified have a near-symmetrical structure with an arm on one side. The arm doesn’t contribute to the plasmonic motion, but it helps anchor the molecule to the lipid bilayer of the cell membrane.”
The researchers had to prove that the molecules’ mode of action could not be categorized either as a form of photodynamic or photothermal therapy.
“What needs to be highlighted is that we’ve discovered another explanation for how these molecules can work,” Ayala-Orozco said. “This is the first time a molecular plasmon is utilized in this way to excite the whole molecule and to actually produce mechanical action used to achieve a particular goal ⎯ in this case, tearing apart cancer cells’ membrane.

“This study is about a different way to treat cancer using mechanical forces at the molecular scale.”
Researchers at Texas A&M University led by Jorge Seminario, a quantum chemist and professor of chemical engineering, performed time-dependent density functional theory analysis on the molecular features involved in the jackhammering effect. The cancer studies were performed in mice at the University of Texas MD Anderson Cancer Center in collaboration with Dr. Jeffrey Myers, professor and chair of the Department of Head and Neck Surgery and director of translational research for the Division of Surgery.
©️ Rice University


























Leave a Reply